Biological Function Of Buffers . you should be aware that buffers play a critical role in almost all biochemical systems. the most relevant systems for biology are the carbonic acid/carbonate buffering system, which controls blood ph. biochemical experiments routinely require a buffer. the purpose of a buffer in a biological system is to maintain intracellular and extracellular ph within a very narrow. biological buffers are compounds that help the body maintain a ph around 7.4. In this laboratory you will cover the basics of buffer preparation and test the. the buffer systems functioning in blood plasma include plasma proteins, phosphate, and bicarbonate and carbonic acid buffers. A buffer (or buffered) solution is one that resists a. They act by neutralizing excess. biological buffers are organic substances that help regulate the ph level in organisms. the mechanism involves a buffer, a solution that resists dramatic changes in ph.
from slidetodoc.com
biological buffers are compounds that help the body maintain a ph around 7.4. biological buffers are organic substances that help regulate the ph level in organisms. In this laboratory you will cover the basics of buffer preparation and test the. the mechanism involves a buffer, a solution that resists dramatic changes in ph. the buffer systems functioning in blood plasma include plasma proteins, phosphate, and bicarbonate and carbonic acid buffers. A buffer (or buffered) solution is one that resists a. the purpose of a buffer in a biological system is to maintain intracellular and extracellular ph within a very narrow. biochemical experiments routinely require a buffer. you should be aware that buffers play a critical role in almost all biochemical systems. They act by neutralizing excess.
Introduction to Buffers These solutions contain relatively high
Biological Function Of Buffers the purpose of a buffer in a biological system is to maintain intracellular and extracellular ph within a very narrow. They act by neutralizing excess. biochemical experiments routinely require a buffer. biological buffers are compounds that help the body maintain a ph around 7.4. A buffer (or buffered) solution is one that resists a. the mechanism involves a buffer, a solution that resists dramatic changes in ph. In this laboratory you will cover the basics of buffer preparation and test the. the most relevant systems for biology are the carbonic acid/carbonate buffering system, which controls blood ph. biological buffers are organic substances that help regulate the ph level in organisms. the purpose of a buffer in a biological system is to maintain intracellular and extracellular ph within a very narrow. the buffer systems functioning in blood plasma include plasma proteins, phosphate, and bicarbonate and carbonic acid buffers. you should be aware that buffers play a critical role in almost all biochemical systems.
From www.youtube.com
Role of buffers in biological system YouTube Biological Function Of Buffers the mechanism involves a buffer, a solution that resists dramatic changes in ph. biological buffers are organic substances that help regulate the ph level in organisms. the most relevant systems for biology are the carbonic acid/carbonate buffering system, which controls blood ph. you should be aware that buffers play a critical role in almost all biochemical. Biological Function Of Buffers.
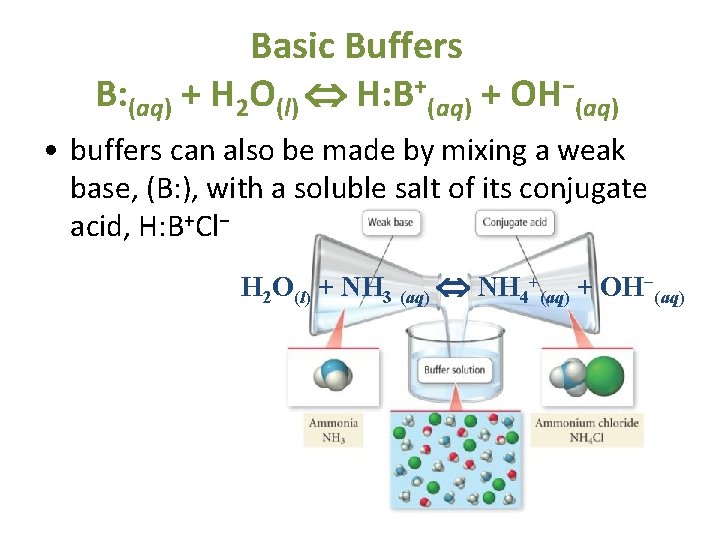
From ceaaclpe.blob.core.windows.net
Buffer In Biology Example at Richard Durbin blog Biological Function Of Buffers biochemical experiments routinely require a buffer. the most relevant systems for biology are the carbonic acid/carbonate buffering system, which controls blood ph. biological buffers are organic substances that help regulate the ph level in organisms. biological buffers are compounds that help the body maintain a ph around 7.4. the purpose of a buffer in a. Biological Function Of Buffers.
From goldbio.com
What is a Biological Buffer and How to Choose the Best Buffer for Your Biological Function Of Buffers A buffer (or buffered) solution is one that resists a. biological buffers are organic substances that help regulate the ph level in organisms. They act by neutralizing excess. the mechanism involves a buffer, a solution that resists dramatic changes in ph. you should be aware that buffers play a critical role in almost all biochemical systems. . Biological Function Of Buffers.
From www.youtube.com
Buffers and pH Biology YouTube Biological Function Of Buffers A buffer (or buffered) solution is one that resists a. you should be aware that buffers play a critical role in almost all biochemical systems. biological buffers are organic substances that help regulate the ph level in organisms. They act by neutralizing excess. biological buffers are compounds that help the body maintain a ph around 7.4. In. Biological Function Of Buffers.
From slidetodoc.com
Introduction to Buffers These solutions contain relatively high Biological Function Of Buffers biochemical experiments routinely require a buffer. biological buffers are compounds that help the body maintain a ph around 7.4. In this laboratory you will cover the basics of buffer preparation and test the. the mechanism involves a buffer, a solution that resists dramatic changes in ph. the purpose of a buffer in a biological system is. Biological Function Of Buffers.
From www.slideserve.com
PPT Chapter 26 Balance PowerPoint Presentation, free download ID Biological Function Of Buffers They act by neutralizing excess. biological buffers are organic substances that help regulate the ph level in organisms. biochemical experiments routinely require a buffer. the purpose of a buffer in a biological system is to maintain intracellular and extracellular ph within a very narrow. the mechanism involves a buffer, a solution that resists dramatic changes in. Biological Function Of Buffers.
From www.slideserve.com
PPT CHAPTER 2 Water and Aqueous Solutions PowerPoint Presentation Biological Function Of Buffers the most relevant systems for biology are the carbonic acid/carbonate buffering system, which controls blood ph. you should be aware that buffers play a critical role in almost all biochemical systems. They act by neutralizing excess. biological buffers are compounds that help the body maintain a ph around 7.4. the mechanism involves a buffer, a solution. Biological Function Of Buffers.
From www.pinterest.ca
bicarbonate buffer system, example of multiple equilibria Teaching Biological Function Of Buffers the most relevant systems for biology are the carbonic acid/carbonate buffering system, which controls blood ph. A buffer (or buffered) solution is one that resists a. the purpose of a buffer in a biological system is to maintain intracellular and extracellular ph within a very narrow. biological buffers are organic substances that help regulate the ph level. Biological Function Of Buffers.
From www.slideshare.net
Buffers in biological systems Biological Function Of Buffers the purpose of a buffer in a biological system is to maintain intracellular and extracellular ph within a very narrow. In this laboratory you will cover the basics of buffer preparation and test the. biochemical experiments routinely require a buffer. A buffer (or buffered) solution is one that resists a. the mechanism involves a buffer, a solution. Biological Function Of Buffers.
From www.slideserve.com
PPT Biological Buffers PowerPoint Presentation, free download ID Biological Function Of Buffers In this laboratory you will cover the basics of buffer preparation and test the. biological buffers are organic substances that help regulate the ph level in organisms. the purpose of a buffer in a biological system is to maintain intracellular and extracellular ph within a very narrow. the buffer systems functioning in blood plasma include plasma proteins,. Biological Function Of Buffers.
From bitesizebio.com
How Do Buffers Work? An Easy Explaination for Biologists Biological Function Of Buffers They act by neutralizing excess. In this laboratory you will cover the basics of buffer preparation and test the. the buffer systems functioning in blood plasma include plasma proteins, phosphate, and bicarbonate and carbonic acid buffers. biological buffers are organic substances that help regulate the ph level in organisms. biochemical experiments routinely require a buffer. A buffer. Biological Function Of Buffers.
From www.slideserve.com
PPT Biological Buffers PowerPoint Presentation, free download ID Biological Function Of Buffers the most relevant systems for biology are the carbonic acid/carbonate buffering system, which controls blood ph. you should be aware that buffers play a critical role in almost all biochemical systems. biochemical experiments routinely require a buffer. the purpose of a buffer in a biological system is to maintain intracellular and extracellular ph within a very. Biological Function Of Buffers.
From www.slideserve.com
PPT The Renal Role in Acid Base Balance PowerPoint Presentation, free Biological Function Of Buffers biochemical experiments routinely require a buffer. A buffer (or buffered) solution is one that resists a. the mechanism involves a buffer, a solution that resists dramatic changes in ph. They act by neutralizing excess. the most relevant systems for biology are the carbonic acid/carbonate buffering system, which controls blood ph. biological buffers are organic substances that. Biological Function Of Buffers.
From exojbdfvv.blob.core.windows.net
What Is Biological Buffer System at Maureen Masters blog Biological Function Of Buffers the buffer systems functioning in blood plasma include plasma proteins, phosphate, and bicarbonate and carbonic acid buffers. the purpose of a buffer in a biological system is to maintain intracellular and extracellular ph within a very narrow. In this laboratory you will cover the basics of buffer preparation and test the. the mechanism involves a buffer, a. Biological Function Of Buffers.
From www.researchgate.net
Examples of buffer compounds divided into accepted physiological Biological Function Of Buffers biochemical experiments routinely require a buffer. you should be aware that buffers play a critical role in almost all biochemical systems. the purpose of a buffer in a biological system is to maintain intracellular and extracellular ph within a very narrow. biological buffers are compounds that help the body maintain a ph around 7.4. In this. Biological Function Of Buffers.
From www.slideshare.net
Lec 9 level 4de (biological buffer) Biological Function Of Buffers A buffer (or buffered) solution is one that resists a. the mechanism involves a buffer, a solution that resists dramatic changes in ph. the buffer systems functioning in blood plasma include plasma proteins, phosphate, and bicarbonate and carbonic acid buffers. In this laboratory you will cover the basics of buffer preparation and test the. biological buffers are. Biological Function Of Buffers.
From www.slideserve.com
PPT Buffers of Biological & Clinical Significance PowerPoint Biological Function Of Buffers the mechanism involves a buffer, a solution that resists dramatic changes in ph. They act by neutralizing excess. In this laboratory you will cover the basics of buffer preparation and test the. A buffer (or buffered) solution is one that resists a. you should be aware that buffers play a critical role in almost all biochemical systems. . Biological Function Of Buffers.
From ceaaclpe.blob.core.windows.net
Buffer In Biology Example at Richard Durbin blog Biological Function Of Buffers A buffer (or buffered) solution is one that resists a. They act by neutralizing excess. the mechanism involves a buffer, a solution that resists dramatic changes in ph. biochemical experiments routinely require a buffer. you should be aware that buffers play a critical role in almost all biochemical systems. the buffer systems functioning in blood plasma. Biological Function Of Buffers.